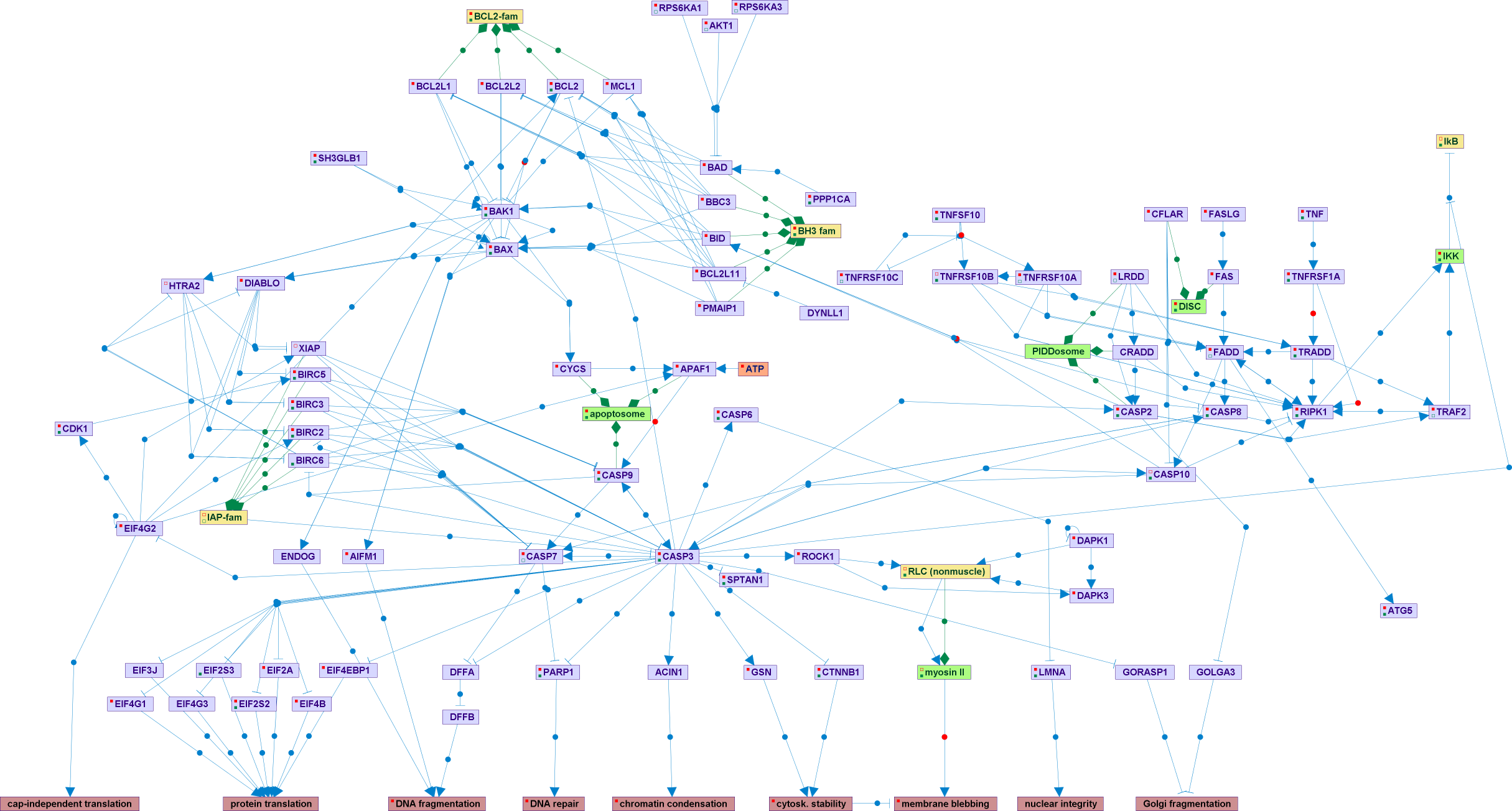
Apoptosis, or type I cell death, is a form of programmed cell death characterized by fragmentation of the cytoplasm and nucleus, chromatin condensation and fragmentation, cytoskeletal collapse, membrane blebbing, and finally, disintegration of the cell into apoptotic bodies, which are engulfed by adjacent cells, thereby avoiding an inflammatory response. It serves to model tissues during embryonic development, to regulate cell number by eliminating excess cells, and to remove damaged, mutated or infected cells. Defective or excessive apoptosis can lead to cancer or autoimmune and degenerative diseases, respectively. Apoptosis is executed by a family of cysteine proteases, the caspases, which function in a proteolytic cascade that is initiated by either the intrinsic mitochondrial-based pathway leading to apoptosome formation, or the extrinsic pathway, which involves activation of death receptors by their extracellular ligands to form the DISC. The most distal caspases, known as the executioner caspases, cleave numerous cellular substrates, thereby leading to specific dismantling of the cell. Apoptosis is regulated by multiple proteins at several levels, including the Bcl-2 family, which regulate the release of apoptogenic factors from the mitochondria, and the IAP family, which regulate caspase activity, and is countered by survival signals eminating from the NF-kB signaling pathway. Although most of the regulation occurs at the post-translational level, involving protein-protein interactions, proteolysis, changes in subcellular localization and phosphorylation, transcriptional control, such as by p53, can also modulate the expression levels of several key apoptotic proteins.